Researchers induce Alzheimer’s neurons from pluripotent stem cells
By By Scott LaFee | 27 Jan 2012
Led by researchers at the University of California, San Diego School of Medicine, scientists have, for the first time, created stem cell-derived, in vitro models of sporadic and hereditary Alzheimer's disease (AD), using induced pluripotent stem cells from patients with the much-dreaded neurodegenerative disorder.
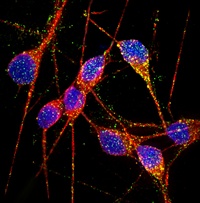 |
| Stem-cell-derived neurons, made from patients with Alzheimer's disease, provide a new tool for unraveling the mechanisms underlying the neurodegenerative disease. In this image, DNA is shown in blue, dendrites and cell bodies in red and endosomal markers Rab5 and EEA1 in green and orange, respectively |
''Creating highly purified and functional human Alzheimer's neurons in a dish – this has never been done before,'' said senior study author Lawrence Goldstein, PhD, professor in the Department of Cellular and Molecular Medicine, Howard Hughes Medical Institute Investigator and director of the UC San Diego Stem Cell Program. ''It's a first step. These aren't perfect models. They're proof of concept. But now we know how to make them. It requires extraordinary care and diligence, really rigorous quality controls to induce consistent behavior, but we can do it.''
The feat, published in the January 25 online edition of the journal Nature, represents a new and much-needed method for studying the causes of AD, a progressive dementia that afflicts approximately 5.4 million Americans. More importantly, the living cells provide an unprecedented tool for developing and testing drugs to treat the disorder.
''We're dealing with the human brain. You can't just do a biopsy on living patients,'' said Goldstein. ''Instead, researchers have had to work around, mimicking some aspects of the disease in non-neuronal human cells or using limited animal models. Neither approach is really satisfactory.''
Goldstein and colleagues extracted primary fibroblasts from skin tissues taken from two patients with familial AD (a rare, early-onset form of the disease associated with a genetic predisposition), two patients with sporadic AD (the common form whose cause is not known) and two persons with no known neurological problems.
They reprogrammed the fibroblasts into induced pluripotent stem cells (iPSCs) that then differentiated into working neurons.
The iPSC-derived neurons from the Alzheimer's patients exhibited normal electrophysiological activity, formed functional synaptic contacts and, critically, displayed tell-tale indicators of AD. Specifically, they possessed higher-than-normal levels of proteins associated with the disorder.


















