New antibodies treat autoimmune disease in mice
28 Dec 2011
A team of Weizmann Institute scientists has turned the tables on an autoimmune disease. In such diseases, including Crohn's and rheumatoid arthritis, the immune system mistakenly attacks the body's tissues. But the scientists managed to trick the immune systems of mice into targeting one of the body's players in autoimmune processes, an enzyme known as MMP9. The results of their research appear in Nature Medicine.
Prof Irit Sagi of the Biological Regulation Department and her research group have spent years looking for ways to home in on and block members of the matrix metalloproteinase (MMP) enzyme family. These proteins cut through such support materials in our bodies as collagen, which makes them crucial for cellular mobilisation, proliferation and wound healing, among other things.
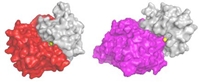 But when some members of the family, especially MMP9, get out of control, they can aid and abet autoimmune disease and cancer metastasis. Blocking these proteins might lead to effective treatments for a number of diseases.
But when some members of the family, especially MMP9, get out of control, they can aid and abet autoimmune disease and cancer metastasis. Blocking these proteins might lead to effective treatments for a number of diseases.
Originally, Sagi and others had designed synthetic drug molecules to directly target MMPs. But these drugs proved to be fairly crude tools that had extremely severe side effects.
The body normally produces its own MMP inhibitors, known as TIMPs, as part of the tight regulation program that keeps these enzymes in line. As opposed to the synthetic drugs, these work in a highly selective manner. An arm on each TIMP is precisely constructed to reach into a cleft in the enzyme that shelters the active bit - a metal zinc ion surrounded by three histidine peptides - closing it off like a snug cork.
''Unfortunately,'' says Sagi, ''it is quite difficult to reproduce this precision synthetically.''
Dr Netta Sela-Passwell began working on an alternative approach as an MSc student in Sagi's lab, and continued on through her PhD research. She and Sagi decided that, rather than attempting to design a synthetic molecule to directly attack MMPs, they would try coaxing the immune system into targeting MMP-9 through immunisation.
Just as immunisation with a killed virus induces the immune system to create antibodies that then attack live viruses, an MMP immunization would trick the body into creating antibodies that block the enzyme at its active site.
Together with Prof. Abraham Shanzer of the Organic Chemistry Department, they created an artificial version of the metal zinc-histidine complex at the heart of the MMP9 active site. They then injected these small, synthetic molecules into mice and afterward checked the mice's blood for signs of immune activity against the MMPs.
The antibodies they found, which they dubbed ''metallobodies,'' were similar but not identical to TIMPS, and a detailed analysis of their atomic structure suggested they work in a similar way - reaching into the enzyme's cleft and blocking the active site. The metallobodies were selective for just two members of the MMP family - MMP2 and 9 - and they bound tightly to both the mouse versions of these enzymes and the human ones.
As they hoped, when they had induced an inflammatory condition that mimics Crohn's disease in mice, the symptoms were prevented when mice were treated with metallobodies.
''We are excited not only by the potential of this method to treat Crohn's,'' says Sagi, but by the potential of using this approach to explore novel treatments for many other diseases.''
Yeda, the technology transfer arm of the Weizmann Institute has applied for a patent for the synthetic immunisation molecules as well as the generated metallobodies.


















