Neutrons provide first sub-nanoscale snapshots of Huntington's disease protein
By By Agatha Bardoel | 06 Jun 2011
Researchers at the Department of Energy's Oak Ridge National Laboratory and the University of Tennessee Medical Center have for the first time successfully characterized the earliest structural formation of the disease type of the protein that causes Huntington's disease.
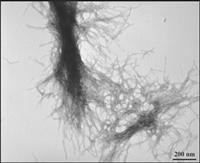 |
| Transmission electron microscopy demonstrates the fibrillar nature of huntingtin aggregates. (Image courtesy C. Stanley, T. Perevozchikova, V. Berthelier, University of Tennessee) |
The incurable, hereditary neurological disorder is always fatal and affects one in 10,000 Americans.
Huntington's disease is caused by a renegade protein "huntingtin" that destroys neurons in areas of the brain concerned with the emotions, intellect and movement. All humans have the normal huntingtin protein, which is known to be essential to human life, although its true biological functions remain unclear.
Christopher Stanley, a Shull Fellow in the Neutron Scattering Science Division at ORNL, and Valerie Berthelier, a UT Graduate School of Medicine researcher who studies protein folding and misfolding in Huntington's, have used a small-angle neutron scattering instrument, called Bio-SANS, at ORNL's High Flux Isotope Reactor to explore the earliest aggregate species of the protein that are believed to be the most toxic.
Stanley and Berthelier, in research published in Biophysical Journal, were able to determine the size and mass of the mutant protein structures -- from the earliest small, spherical precursor species composed of two (dimers) and three (trimers) peptides -- along the aggregation pathway to the development of the resulting, later-stage fibrils. They were also able to see inside the later-stage fibrils and determine their internal structure, which provides additional insight into how the peptides aggregate.
"Bio-SANS is a great instrument for taking time-resolved snapshots. You can look at how this stuff changes as a function of time and be able to catch the structures at the earliest of times," Stanley said. "When you study several of these types of systems with different glutamines or different conditions, you begin to learn more and more about the nature of these aggregates and how they begin forming."


















