New wearable kidney may be boon for dialysis patients
04 Jun 2016
A wearable artificial kidney may be a viable dialysis technology that can give people with end-stage kidney disease the freedom to move around during treatment, according to the results of a new clinical trial.
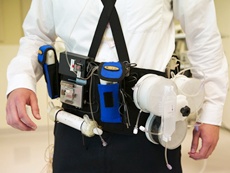 The technology may become an alternative to conventional haemodialysis for people with end-stage kidney disease.
The technology may become an alternative to conventional haemodialysis for people with end-stage kidney disease.
Present-day treatment generally requires three sessions a week on a stationary machine that restricts patients' ability to walk around while it is attached and running.
In contrast, a wearable device would allow patients to be mobile and untethered. It could also provide additional treatment benefits from longer sessions or more frequent days of dialysis.
The clinical trial of a prototype for such a device was performed with seven patients at University of Washington Medical Centre in the US last year. The patients were treated with the device for up to 24 hours.
The US Food and Drug Administration-authorised trial was conducted to determine the safety and efficacy of the device - its ability to take over some functions of failed kidneys.
The researchers also wanted to ask the participants about their impressions of the experimental treatment, and to compare those with standard dialysis treatment.
The device was shown to effectively clear the blood of waste products, like urea, creatinine and phosphorus, while also removing excess water and salt. These are normally filtered out and removed by working kidneys.
The usual diet for patients on standard dialysis is highly limited. In contrast, on the wearable artificial kidney, the patients' blood electrolytes - like sodium and potassium - and their blood fluid volume remained balanced during the test, even without any diet restrictions.
Regulating the volume and composition of body fluids is another job of normal kidneys.
During the trial, the participants tolerated the treatment well and did not have any serious, adverse effects.
The circulatory system, which keeps blood moving throughout the body, stayed stable in all the patients.
However, the trial was stopped after the seventh patient because of technical problems with the device. These included the excessive formation of carbon dioxide gas bubbles in the dialysis solution, and intermittent variations in solution and blood flow.
These technical complications will need to be addressed through device redesign and refinement to enhance safety and reliability prior to any further, long-term studies of the wearable artificial kidney, researchers said.
The findings provide proof of concept that a wearable device along these lines could be developed as a viable, novel dialysis technology, they said.
The patients participating in the study reported greater satisfaction during their treatment with the wearable artificial kidney when compared to their ratings of care during conventional dialysis centre treatment.
The findings were published in the journal JCI Insights.


















