Vaccines hold hope in fight against malaria
25 Apr 2013
The last decade has produced significant gains in the fight against malaria. The number of cases of the disease have dropped globally and so too have the number of deaths. However, the disease still kills an estimated 660,000 people every year, mostly children in Africa.
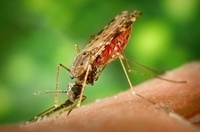 |
| Mosquitoes from the genus Anopheles transmit the protozoan that causes malaria. Credit: Wikimedia |
On World Malaria Day, World Radio Switzerland's international Geneva correspondent, Vincent Landon, took a look at how the war on malaria was progressing and why new hope was vested in vaccines.
Vaccines have enjoyed phenomenal success in reducing and eliminating many diseases and the hope is to replicate that success for malaria.
One vaccine RTS, S had shown the most promising results so far as seen from tests in thousands of children. It had proved to be effective in about 35 per cent of cases, which was though not particularly convincing but if a disease that killed one child every minute, a vaccine which was 35 per cent effective could save a third of them.
According to Landon $2 billion annually was going into the fight against malaria, which was up 10-fold in a decade. He adds that long-lasting insecticide treated nets, indoor residual spraying, diagnostic tests and treatment with artemesinin-based combination therapy had all helped turn the tide.
According to Robert Newman, director of the Global Malaria Programme at the World Health Organization in Geneva, the scale up of these interventions had saved over a million lives in the past decade.
He said that was reflected in a greater than 25 per cent drop in malaria mortality rates globally and over 33 per cent drop in malaria mortality rates in the African region.
He added, however that there were still an estimated 660,000 people a year dying from malaria, mostly African children under the age of five and for a disease that was entirely preventable and treatable, this was totally unacceptable.
Meanwhile, the US Food and Drug Administration today announced a public-private partnership to help identify counterfeit or substandard anti-malarial medicines, including falsified products, with the deployment of the FDA-developed Counterfeit Detection Device, called CD-3.
The FDA said in a press release, "Malaria kills more than 660,000 people globally each year, mostly children. The threat of drug resistance, limited availability of medication, and increased distribution of counterfeit or substandard anti-malarial medicines pose significant challenges to treating this disease.
"Compromised anti-malarial medicines often have too little or no active ingredients, preventing adequate and timely treatment. Anti-malarial medicines made with reduced dosages of active ingredients will not cure patients with malaria, and they can lead to resistant strains of the parasite, making it tougher to treat malaria, even with authentic medicines.
To minimise patient exposure to this public health threat, the FDA has established a partnership with the Skoll Global Threats Fund, the US Pharmacopeia (USP), the National Institutes of Health (NIH), the Centers for Disease Control and Prevention (CDC), and the multi-agency President's Malaria Initiative (PMI), led by the US Agency for International Development (USAID).
The partnership will focus on testing and optimising the use of the handheld CD-3 to identify counterfeit or substandard anti-malarial medicines, including falsified products, in Africa and parts of Southeast Asia, where the rates of malaria infection are high and where counterfeit anti-malarial medicines are prevalent.
FDA also announced today it has signed a letter of intent with Corning Incorporated to refine and improve the tool for eventual manufacture on a larger scale.
''Fake or substandard anti-malarial drugs cause double damage: without adequate, prompt treatment, the malaria parasite can kill a person in a matter of days, and inadequate treatment can also lead to the development of drug resistance, potentially rendering all treatment ineffective,'' said FDA commissioner Margaret A Hamburg, MD. ''The development of the CD-3 and the formation of this important partnership are critical steps toward the FDA's goal of improving the global product safety net in order to protect consumers in the U.S. and worldwide.''
The effectiveness of the tool in detecting counterfeit or substandard versions of two common anti-malarial therapies will be tested in Ghana in 2013 and 2014. The USP Promoting the Quality of Medicines Program (PQM), with funds from USAID and PMI, collaborates with the Ghanaian Food and Drug Authority to conduct drug surveillance programs at test sites in Ghana, and the new partnership will leverage this existing infrastructure. CDC and NIH will provide technical support, and The Skoll Global Threats Fund will provide additional funding for the initial testing program in Ghana.
Information obtained from the test in Ghana is intended to guide a second testing program, which will also make use of existing infrastructure from the PQM program.
In a recent report commissioned by the FDA, the Institute of Medicine concluded that making detection technology more accessible to low and middle income countries would be invaluable in controlling the trade in counterfeit, falsified, or substandard medicines.
''The proliferation of counterfeit or substandard medicines around the world is a major public health problem and the developing world is disproportionately affected,'' said commissioner Hamburg. ''The FDA is pleased that CD-3 has the potential to be used as a frontline tool in protecting the global supply chain and make a positive impact on public health efforts to combat malaria in developing regions of the world that are particularly hard hit by the disease.''
The CD-3 is a handheld, battery-operated tool that illuminates a product with a variety of wavelengths of light to provide a visual comparison of an unverified product with an authentic sample. This allows inspectors to identify suspect products and remove them from the supply chain. Minimal scientific or technical background is needed to operate the tool, and it can be used even in remote communities or in places with only very basic health care systems.
Scientists at the FDA's Forensic Chemistry Center in Cincinnati, Ohio, developed the tool, which has been in use since 2010 in their laboratory as well as in a number of international mail facilities in the US and other points of entry where investigators screen drug ingredients and other finished products to identify counterfeit or unapproved products. The tool has also been applied effectively to screen cosmetics, foods, medical devices, and cigarettes, and investigate product tampering and questionable documents.




















